What Are Primer and Probe in Molecular Biology?
Primer and probe are fundamental molecular tools that have transformed how scientists detect, amplify, and analyze DNA sequences. These short synthetic DNA or RNA fragments serve as essential components in various laboratory techniques, particularly in polymerase chain reaction (PCR) and quantitative PCR (qPCR) applications.
Understanding primer and probe technology is crucial for anyone working in genetics, diagnostics, or research. Whether you’re detecting infectious diseases, identifying genetic mutations, or conducting forensic analysis, these molecular instruments play a vital role in obtaining accurate and reliable results.
Understanding Primers: The Building Blocks of DNA Amplification
What Is a Primer?
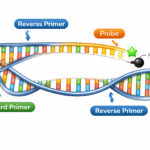
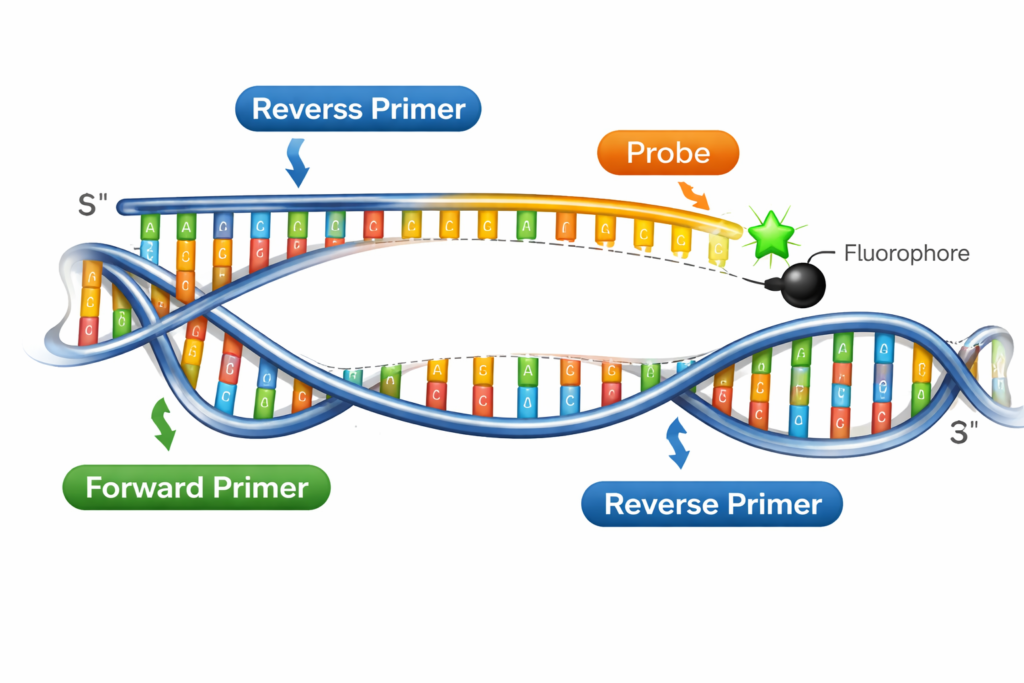
A primer is a short, single-stranded nucleic acid sequence, typically 18-25 nucleotides long, designed to bind to a specific complementary DNA sequence. Primers serve as starting points for DNA synthesis, allowing DNA polymerase enzymes to begin copying a target DNA sequence.
In any PCR reaction, you need two primers:
Forward primer: Binds to the 3′ end of the antisense strand
Reverse primer: Binds to the 3′ end of the sense strand
These primers flank the target DNA region you want to amplify, ensuring specific and accurate replication.
How Primers Work
The primer and probe mechanism begins when primers anneal (attach) to their complementary sequences on the template DNA. Once bound, DNA polymerase recognizes the primer-template junction and starts synthesizing new DNA strands.
This process is temperature-dependent and occurs during the annealing phase of PCR, typically at temperatures between 50-65°C. The specificity of primer binding is critical for successful amplification of the desired DNA segment.
Key Characteristics of Effective Primers
Well-designed primers should have:
– GC content between 40-60%
– Melting temperature (Tm) between 52-68°C
– Minimal self-complementarity to avoid primer-dimer formation
– No secondary structures like hairpins
– Specific binding to only the target sequence
Understanding Probes: The Detection Powerhouses
What Is a Probe?
A probe is a single-stranded DNA or RNA sequence, typically 20-30 nucleotides long, designed to bind to a specific complementary sequence within the target DNA. Unlike primers, probes are labeled with fluorescent dyes or other markers, enabling real-time detection and quantification of DNA amplification.
The primer and probe combination creates a powerful system for both amplifying and detecting specific DNA sequences simultaneously.
Types of Probes
Several probe technologies are commonly used:
1. Hydrolysis Probes (TaqMan Probes):
These probes contain a fluorescent reporter dye at one end and a quencher molecule at the other. When the probe is intact, fluorescence is suppressed. During PCR amplification, the DNA polymerase cleaves the probe, separating the reporter from the quencher and generating a fluorescent signal.
2. Hybridization Probes (FRET Probes):
These consist of two oligonucleotides that bind adjacently to the target sequence. When both probes hybridize, energy transfer occurs between fluorophores, producing a measurable signal.
3. Molecular Beacon Probes:
These hairpin-shaped probes contain complementary sequences at their ends. Upon binding to the target, the hairpin opens, separating the fluorophore from the quencher and emitting fluorescence.
4. Scorpion Probes:
These self-probing primers contain the primer sequence linked to a probe element, offering enhanced specificity and faster detection.
Primer and Probe Applications in Real-World Settings
- Disease Diagnosis and Detection
The primer and probe technology has revolutionized medical diagnostics. Healthcare professionals use these tools to:
– Detect viral infections like COVID-19, HIV, and hepatitis
– Identify bacterial pathogens in clinical samples
– Diagnose genetic disorders and inherited diseases
– Monitor viral load in patients undergoing treatment
According to the [World Health Organization](https://www.who.int/), molecular diagnostic tests using primer and probe systems have become the gold standard for many infectious disease detections.
2. Genetic Research and Analysis
Researchers employ primer and probe techniques for:
– Gene expression studies
– Single nucleotide polymorphism (SNP) detection
– Copy number variation analysis
– Epigenetic modifications assessment
– Mutation screening in cancer research
3. Food Safety and Quality Control
The food industry utilizes primer and probe technology to:
– Detect foodborne pathogens like Salmonella and E. coli
– Verify food authenticity and prevent fraud
– Identify genetically modified organisms (GMOs)
– Monitor allergen presence in food products
4. Forensic Science
Forensic laboratories depend on primer and probe systems for:
– DNA fingerprinting and identification
– Paternity testing
– Crime scene analysis
– Human remains identification
5. Environmental Monitoring
Environmental scientists use these molecular tools to:
– Detect microbial contamination in water sources
– Monitor biodiversity through environmental DNA (eDNA)
– Track invasive species
– Assess ecosystem health
The Primer and Probe Workflow in qPCR
Understanding how primer and probe work together in quantitative PCR helps appreciate their importance:
Step 1: Denaturation
The reaction mixture is heated to 94-95°C, causing double-stranded DNA to separate into single strands.
Step 2: Annealing
Temperature drops to 50-65°C, allowing primers and probes to bind to their complementary sequences on the template DNA.
Step 3: Extension
At 72°C, DNA polymerase synthesizes new DNA strands starting from the primers. If a probe is present, it gets cleaved during this process, generating a fluorescent signal.
Step 4: Detection
The fluorescence released is measured in real-time, allowing for quantification of the initial DNA amount.
This cycle repeats 25-40 times, exponentially amplifying the target sequence while providing quantitative data through probe fluorescence
Designing Effective Primer and Probe Sets
Critical Design Considerations
Creating optimal primer and probe combinations requires careful planning:
For Primers:
– Ensure primers are complementary to the target sequence
– Avoid sequences with repetitive elements
– Check for potential off-target binding using BLAST searches
– Maintain similar Tm values for both forward and reverse primers
– Design primers that produce amplicons between 70-150 base pairs for qPCR
For Probes:
– Position the probe between the forward and reverse primers
– Ensure probe Tm is 5-10°C higher than primer Tm
– Select appropriate fluorophore-quencher combinations
– Verify probe doesn’t form secondary structures
– Confirm no overlap with primer binding sites
Software Tools for Design
Several bioinformatics tools assist in primer and probe design:
– Primer3 and Primer-BLAST for primer design
– Beacon Designer for probe optimization
– OligoAnalyzer for secondary structure prediction
– IDT PrimerQuest for integrated design solutions
Advantages of Using Primer and Probe Technology
The primer and probe system offers numerous benefits:
1. High Specificity
The dual-binding requirement (primers for amplification, probe for detection) ensures exceptional specificity, reducing false-positive results.
2. Real-Time Monitoring
Unlike conventional PCR, primer and probe-based qPCR allows real-time monitoring of amplification, providing quantitative data.
3. Multiplexing Capability
Multiple primer and probe sets with different fluorophores can detect several targets in a single reaction, saving time and resources.
4. Wide Dynamic Range
These systems can accurately quantify DNA across several orders of magnitude, from a few copies to millions.
5. Reduced Contamination Risk
Closed-tube detection eliminates the need for post-PCR analysis, minimizing contamination risks.
6. Automation-Friendly
Primer and probe assays are easily automated, enabling high-throughput screening in clinical and research laboratories.
Common Challenges and Troubleshooting
Primer-Dimer Formation
This occurs when primers bind to each other instead of the template. Solutions include:
– Redesigning primers to minimize complementarity
– Optimizing primer concentrations
– Adjusting annealing temperatures
– Using hot-start polymerases
Non-Specific Amplification
When primers amplify unintended sequences:
– Increase annealing temperature
– Redesign primers for better specificity
– Optimize magnesium ion concentration
– Use touchdown PCR protocols
Probe Degradation
Improper storage or handling can degrade probes:
– Store probes at -20°C in the dark
– Minimize freeze-thaw cycles
– Use nuclease-free reagents and consumables
– Verify probe integrity before use
Low or No Signal
Troubleshooting weak signals:
– Check probe and primer concentrations
– Verify correct fluorophore-quencher pairing
– Ensure template DNA quality and quantity
– Confirm proper instrument calibration
Future Trends in Primer and Probe Technology
The field continues to evolve with exciting developments:
1. Digital PCR
Combining primer and probe technology with digital PCR enables absolute quantification without standard curves, offering enhanced precision.
2. CRISPR-Based Detection
Integrating CRISPR technology with primer and probe systems creates ultrasensitive detection platforms for diagnostics.
3. Microfluidics Integration
Miniaturized systems using primer and probe assays enable point-of-care testing with minimal sample requirements.
4. AI-Assisted Design
Machine learning algorithms improve primer and probe design, predicting performance and optimizing assay conditions.
5. Enhanced Fluorophores
Novel fluorescent dyes with improved brightness and photostability enhance detection sensitivity and multiplexing capabilities.
According to the [National Center for Biotechnology Information](https://www.ncbi.nlm.nih.gov/), these advances continue to expand the applications of primer and probe technology in personalized medicine and precision diagnostics.
Selecting the Right Primer and Probe for Your Application
Consider Your Specific Needs
Different applications require different approaches:
For Clinical Diagnostics:
– Prioritize sensitivity and specificity
– Choose FDA-approved or CE-marked assays when possible
– Ensure robust performance across sample types
– Select probes with stable fluorophores
For Research Applications:
– Balance cost with performance requirements
– Consider multiplexing needs
– Evaluate turnaround time for custom designs
– Assess compatibility with existing equipment
For High-Throughput Screening:
– Opt for automation-compatible formats
– Choose cost-effective options for large volumes
– Ensure batch-to-batch consistency
– Consider pre-designed, validated assay kits
Working with Suppliers
When ordering custom primer and probe sets:
– Provide complete sequence information
– Specify modification requirements clearly
– Request quality control certificates
– Inquire about technical support availability
– Compare pricing across reputable suppliers
Leading suppliers include [Thermo Fisher Scientific](https://www.thermofisher.com/), Integrated DNA Technologies (IDT), and Sigma-Aldrich, all offering comprehensive primer and probe services.
Best Practices for Primer and Probe Storage and Handling
Storage Guidelines
Proper storage extends the shelf life of your primer and probe reagents:
– Store lyophilized primers and probes at -20°C in desiccated conditions
– Once reconstituted, aliquot into single-use portions
– Avoid repeated freeze-thaw cycles
– Protect probes from light exposure
– Maintain detailed inventory records with expiration dates
Handling Recommendations
– Always use nuclease-free water for reconstitution
– Wear gloves to prevent contamination
– Work in a clean, dedicated PCR setup area
– Centrifuge tubes briefly before opening
– Use calibrated pipettes for accurate measurements
Conclusion:
The Indispensable Role of Primer and Probe
Primer and probe technology has fundamentally transformed molecular biology, diagnostics, and research. These molecular tools provide the specificity, sensitivity, and quantitative capabilities needed for modern genetic analysis.
From detecting emerging infectious diseases to advancing personalized medicine, primer and probe systems continue to be at the forefront of scientific innovation. As technology advances, their applications will only expand, making them even more essential in laboratories worldwide.
Whether you’re a researcher designing new assays, a clinician interpreting diagnostic results, or a student learning molecular techniques, understanding primer and probe technology is crucial. By mastering these fundamental tools and following best practices in design, implementation, and troubleshooting, you can harness their full potential for your specific applications.
The future of molecular diagnostics and research depends on these remarkable molecular instruments. As we continue to face global health challenges and push the boundaries of genetic understanding, primer and probe technology will remain an indispensable part of our scientific toolkit
Frequently Asked Questions
What's the main difference between a primer and a probe?
Primers initiate DNA synthesis and are used in pairs to amplify target sequences, while probes are fluorescently labeled oligonucleotides used to detect and quantify specific DNA sequences during amplification.
Can I use the same primer and probe set for different samples?
Yes, if the target sequence is conserved across samples. However, always validate performance with each new sample type to ensure specificity and sensitivity.
How long do primer and probe reagents last?
When properly stored at -20°C, lyophilized primers can last 1-2 years, while probes typically remain stable for 6-12 months. Reconstituted solutions have shorter shelf lives.
Are primer and probe assays expensive?
Costs vary depending on modifications, scale, and supplier. Standard primers are relatively inexpensive, while labeled probes cost more due to fluorophore additions. However, the accuracy and efficiency often justify the investment.
Medical Disclaimer:
This article is for educational purposes only and does not replace professional medical advice. Always follow the specific instructions provided by your healthcare provider or laboratory.